Selling pet food (for dogs and cats) involves labeling rules that differ from human food. In Japan, pet food packages must clearly display—in Japanese—the “five mandatory labeling items” stipulated by law. In addition, industry standards based on the Fair Competition Code require further information that is essential in practice, such as the product’s intended purpose (e.g., “complete and balanced”), guaranteed analysis values, and feeding instructions.
This article provides a thorough, practical explanation of: the mandatory labeling items required under the Pet Food Safety Act and their details; labeling items required under the Fair Competition Code; metabolizable energy (ME) labeling (voluntary but practically important); Japanese labeling for imported products; rules for country-of-origin labeling; prohibited/NG expressions; and example formats and checklists for label creation.
Because the key points are summarized under short, clearly separated headings, this guide is intended to be directly useful for professionals responsible for pet food labeling and packaging in day-to-day work.
The “five mandatory labeling items” under the Pet Food Safety Act
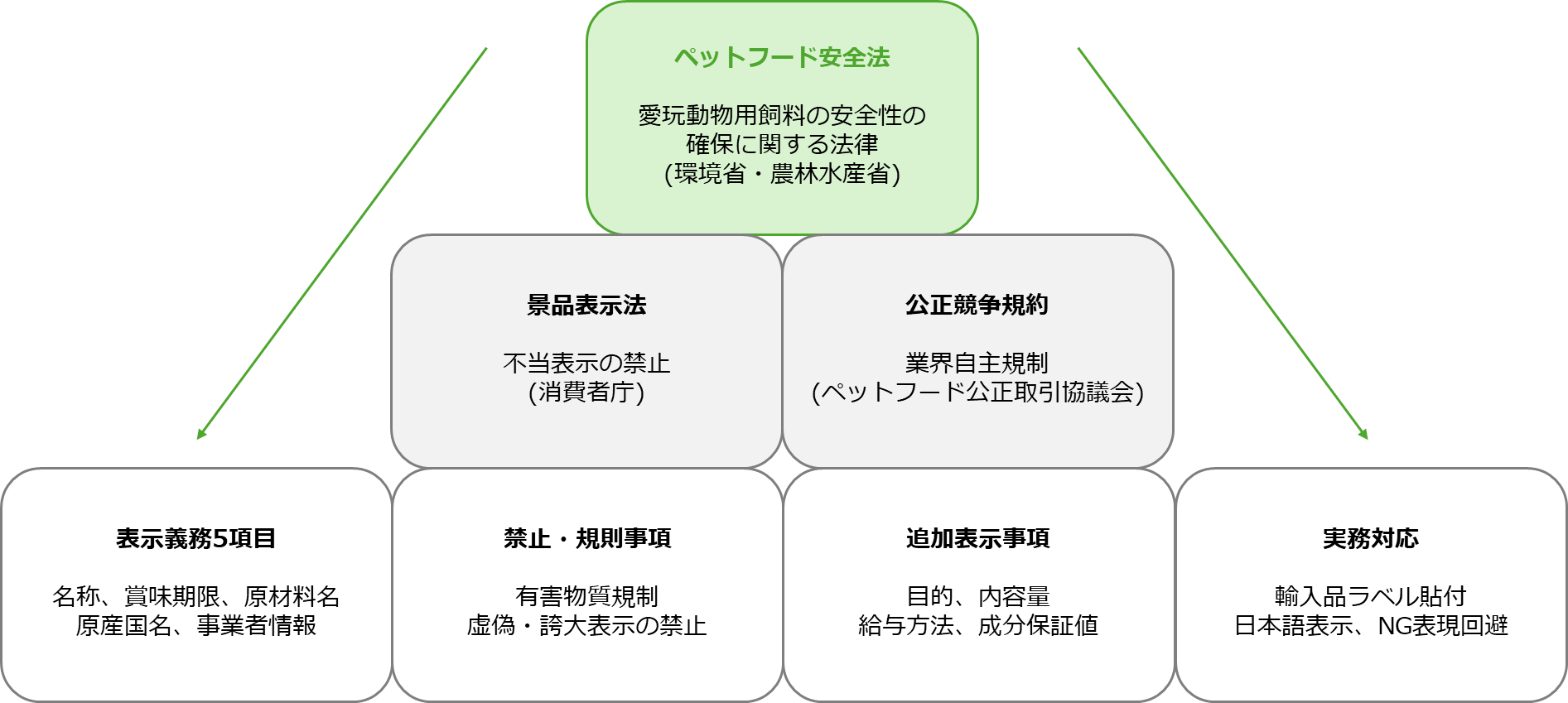
Pet food labeling is governed by multiple laws and industry rules. In practice, those responsible for labeling must understand the overall framework and comply accordingly.
What is the Pet Food Safety Act?
- Official name: Act on Securing Safety of Feed for Pet Animals
- Purpose: To ensure the safety of pet food, protect the health of companion animals, and contribute to animal welfare
- Effective date: June 1, 2009 (Heisei 21)
- Competent authorities: Ministry of the Environment and Ministry of Agriculture, Forestry and Fisheries (joint jurisdiction)
- Covered animals: Dogs and cats (currently limited to these animals under the Act)
Key characteristics of the Pet Food Safety Act
- Mandatory labeling of the five required items
- Regulation of hazardous substances
- Notification (filing) obligation for manufacturers and importers
- Government monitoring and administrative measures/orders
Penalties for labeling violations
Violations of the labeling obligations are punishable by imprisonment for up to one year and/or a fine of up to 1,000,000 yen.
Mandatory labeling under the Pet Food Safety Act
Pet food is subject to the Act on Securing Safety of Feed for Pet Animals (commonly known as the Pet Food Safety Act), which defines required labeling items.
The Food Labeling Act for human food does not apply to pet food. Instead, the Pet Food Safety Act (enforced in 2009) provides the applicable standards. Under this Act, pet food sold in Japan must display the following five items in Japanese on the package:
Below, each item is explained with practical points for day-to-day labeling work.
1. Name: product name and intended animal category
“Name” refers to the product name and the indication of whether it is for dogs and/or cats. The front of the package must make it obvious at a glance whether the food is for dogs or cats.
For example, clearly state “○○ dog food” or “△△ cat food,” separate from the brand/product name if needed. In addition, include “for dogs” or “for cats” again in the consolidated labeling panel (detailed label area). If the product is suitable for both, it must be labeled “for dogs and cats.”
Examples
You may add wording that clarifies the target animal and life stage, such as “Adult dog food ○○” or “Kitten nutritional supplement.” If the product can also be fed to animals other than dogs/cats, you must still clearly indicate the dog/cat applicability, e.g., “for dogs, cats, and ○○.”The name is often the first information consumers notice. Legally, it is sufficient to indicate “for dogs” or “for cats,” but in practice many brands include life stage (puppy, adult, senior) and use/purpose for clarity. For example, “Complete and balanced for adult dogs ○○” or “Weight management cat food △△” helps consumers understand the target and purpose immediately.
2. Best-before date: clear year/month(/day) indication
The best-before date indicates the date until which the product can be fed while maintaining quality and palatability. It must be shown as year/month/day or year/month, in that order, using Arabic numerals.
Examples: “October 31, 2025” or “October 2025.” (If only year/month is shown, the best-before date is the last day of that month.) Display it clearly together with a label such as “Best before:”.
If best-before information is coded (e.g., letters/numbers printed on the seal area), it is recommended to add a consumer-friendly explanation such as: “In the LOT number, ‘2306’ indicates June 2023,” or “Best-before is printed on the bottom in day/month/year order,” to avoid confusion.
You may also voluntarily display the manufacturing date or use-by date in addition to the best-before date. In all cases, because best-before is critical for safety and quality assurance, it should be easy for consumers to find and read.
3. Ingredients: list all ingredients (including additives)
The ingredients section must list all ingredients used in the product, without omission, including additives such as preservatives and colorants. The law does not explicitly mandate the order, but in practice it is generally recommended to list ingredients from highest to lowest inclusion level.
When labeling in accordance with the Fair Competition Code, the recommended format is: list non-additive ingredients in descending order by use amount, then list additives together.
When using additives for specific functions—such as colorants, preservatives, or antioxidants—labeling should include the additive name and its function/purpose, for example: “Antioxidant (mixed tocopherols).”
Certain processing aids (substances used during manufacturing that do not remain in the final product) may be exempt from labeling in some cases. However, exemptions are only allowed when supported by scientifically reasonable grounds. Be careful so that it is not considered an omission.
Also note the handling of vitamin/mineral premixes. It is not acceptable to label them simply as “vitamin mix,” etc. Each vitamin and mineral must be listed individually (e.g., “Vitamin A, Vitamin D, calcium, zinc…”).
The ingredient list is a key source of information for consumers assessing what the product contains. Even when you want to emphasize differentiation (e.g., “made with ○○ chicken,” “gluten-free”), you must label accurately within the legal framework.
If you highlight ingredient origin or quality claims, avoid misleading wording (unfairly superior claims). Use fact-based expressions that do not create misunderstanding, such as “Contains X% ○○ chicken from ○○,” only where accurate, and keep the risk of misleading representation in mind (explained later).
4. Country of origin: label the country of final processing
The country of origin is the country where the final processing step for the pet food was completed. For imported products, the origin is not the country where ingredients were sourced, but the country where the product was finished as a product.
Label as “Country of origin: ○○.” If it is manufactured in Japan, it may be labeled as “Japan” or “Made in Japan” (domestic). For example, even if the ingredients are from the U.S., if the food is processed and packed in Thailand, the country of origin should be “Thailand.”
Country-of-origin labeling strongly affects perceived reliability and safety, so using other countries’ names in ways that could mislead is strictly prohibited.
For example, placing a large foreign flag or map of a different country on the package, or using place names associated with a country other than the actual origin (“Paris” when not made in France) can create the risk of misleading consumers.
The Fair Competition Code also specifies that you must not use labeling that could mislead consumers about the origin beyond the origin statement itself. Even when promoting the use of overseas ingredients, keep the presentation consistent with the actual country-of-origin labeling.
5. Business operator name and address: clearly identify the responsible party
Packages must display the business operator’s name (or individual name) and address. This ensures that, in case of problems, responsibility is clear and allows swift investigation and recall.
In practice, it is recommended to clarify the operator type—manufacturer, importer, or distributor—for example:
- “Manufacturer: ○○ Co., Ltd. (address…)”
- “Importer: △△ Trading Co., Ltd. (address…)”
Avoid vague terms such as “manufactured by” or “distributed by” in an informal way; use formal role designations such as “manufacturer,” “importer,” or “distributor/seller,” as appropriate. Because the legal obligation is “name and address,” the address must be included—not just the company name.
For imported products, it is common to list the importer responsible for distribution in Japan. You may also list the overseas manufacturer, but it is not mandatory. Do not abbreviate the address; include details accurately, including building name and unit/room number where applicable.
Example
- Manufacturer: ○○ Pet Food Co., Ltd.
1-2-3 ○○, Chiyoda-ku, Tokyo, ○○ Building 5F - (Not recommended) “Maker: ○○ Pet Food (Co.)” / “Tokyo, Chiyoda-ku …” (abbreviated)
These are the five mandatory items under the Pet Food Safety Act. It is generally recommended to group them into one consolidated labeling area. Even with limited package space, layout should be designed for clarity and readability. Next, we explain additional items under the “Fair Competition Code” and other practical voluntary information.
The “four labeling items” under the Fair Competition Code
The pet food industry in Japan has a self-regulatory standard called the “Fair Competition Code Concerning Labeling of Pet Food” (approved by the Japan Fair Trade Commission and the Consumer Affairs Agency).
Member companies of the Pet Food Fair Trade Council comply with this Code and, in addition to the five legal items, they label four further items: purpose, net content, feeding method, and composition (guaranteed analysis).
Even for non-member companies, these items are not legally mandatory, but they are essentially standard in practice because they provide consumers with useful information and are commonly expected in the marketplace.
Purpose: intended use category
Under the Code, pet food products should display the product’s “purpose” using one of the following four categories:
This indicates how the product should be positioned (staple diet, treat, etc.), enabling consumers to understand its role correctly.
Complete and balanced
A diet designed to provide all essential nutrients necessary to maintain health for a specific life stage when fed with water. It must specify the applicable life stage (puppy/kitten, adult, senior, or “all life stages”).
To label a product “complete and balanced,” it must meet nutritional standards (e.g., AAFCO nutrient profiles) and the compliance must be supported via analysis and/or feeding trials. Some labels also include a statement that the product meets the “complete and balanced” standards for a specified life stage.
Snacks/treats
Supplemental foods fed as rewards or treats, such as jerky, biscuits, or freeze-dried treats. Because these are not staple foods and are often palatability-focused, it is also advisable to include guidance to avoid overfeeding.
Therapeutic diet
A diet intended to support treatment or management of specific diseases under veterinary guidance. Because this category differs from general pet food, special care is needed with labeling (e.g., statements such as “Feed only under the guidance of a veterinarian”). Note that labeling therapeutic diets may require notification/filing with the Fair Trade Council and must remain within acceptable labeling boundaries, including considerations related to pharmaceutical regulations.
Other purpose foods
Foods intended for purposes other than the above, such as complementary foods, side dishes, or topping-type products that are not complete and balanced. Such products should indicate that they are to be fed together with other foods (e.g., “Feed in combination with complete and balanced food”). Examples include “complementary food,” “side dish,” or “supplementary food.”
This purpose labeling is critical—especially “complete and balanced,” which signals that the diet is nutritionally complete as the main meal. If a product that does not meet the standards is labeled in a way that could be interpreted as a staple diet, it risks being misleading.
Net content: display net weight or quantity
Net content (net amount) is also required under the Code. It should be shown in weight (g/kg) or volume (ml/L), as appropriate.
For multi-packs, displaying both the total and breakdown is helpful (e.g., “100 g × 10 packs”). For treats, quantity-based labeling such as “○ sticks” or “○ pieces” is also permitted.
Net content labeling also relates to measurement laws and accurate quantity control. Carefully ensure that actual contents match the labeled amount and do not fall below it due to lot variation. Overly empty packaging designed to make the content appear larger can be considered misleading.
Feeding method: appropriate daily feeding amount and frequency
Feeding method labeling provides guidance on how much and how often to feed per day. Because energy needs vary by age, body weight, and activity level, labels typically present model feeding amounts and recommended feeding frequency.
This can be written as text, presented in a body-weight table, or supported with illustrations. The key is clarity so consumers can understand the appropriate amount intuitively.
Examples
“For an adult dog weighing 5 kg, feed approximately 100 g per day divided into two meals.”
“Daily feeding guide by body weight: see table.”
For puppies, labels often add instructions such as “Soak before feeding,” etc.Although feeding method is not legally mandatory, it is essential to prevent feeding errors and protect animal health—especially for complete and balanced diets, where incorrect feeding can cause over- or under-nutrition. For weight-management products, combining feeding guidance with calorie/ME information is especially useful. Including general tips such as “Provide fresh water at all times” can also be beneficial.
Guaranteed analysis: nutrient composition values
nder the Code, key nutrient composition values (guaranteed analysis) should be displayed as weight percentages for the following five items:
Be careful to use “or more” for protein and fat, and “or less” for fiber, ash, and moisture.
Example: “Crude protein 25% min, crude fat 15% min, crude fiber 4% max, crude ash 8% max, moisture 10% max.”
These five items are basic indicators for evaluating nutritional content. For complete and balanced foods, values must fall within appropriate ranges and the product must meet the applicable standards (e.g., AAFCO nutrient profiles). Product developers should confirm via testing that the analysis supports the declared values and compliance. Significant deviation can cause regulatory and consumer protection issues and may risk animal health.
Voluntary item: metabolizable energy (ME) labeling
Metabolizable energy (ME) refers to the usable energy in pet food—i.e., calories—per kg or per 100 g. In Japan, calorie/energy labeling is not a legal requirement and is voluntary. However, in current practice it is commonly displayed on most products and is effectively an essential piece of information.
In Europe and the U.S., calorie labeling is already required (for example, AAFCO requires energy information for pet food). In Japan, while not formally mandatory, energy information is indispensable for setting feeding amounts and comparing products.
ME can be calculated using AAFCO-accepted formulas (e.g., modified Atwater factors) or determined via measurement. Developers typically estimate calories from guaranteed analysis values and/or confirm with testing, then reflect the result in labeling.
Common formats include “Metabolizable energy (ME): ○○ kcal/100 g.” For single-serve packs or treats, “○ kcal per pouch (○ g)” or “○ kcal per piece” may be more appropriate.
Language requirements and handling for imported products
All the mandatory items and Code-based labeling items described above must be displayed in Japanese. Even if the product is manufactured overseas, if it is sold in Japan, required labeling must be provided in Japanese to consumers.
For imported pet food, the original foreign-language packaging alone is not sufficient. A Japanese label (sticker) that covers the required items must be affixed before sale.
In practice, imported-product labeling requires particular attention to:
Ensure the label does not peel off
Clean/degrease the surface of bags/cans and use strong adhesive labels so they do not peel during transport or on store shelves. A peeled label effectively becomes an unlabeled product and can constitute a violation.
Avoid contradictions with the original label
If the original package already contains information in English, etc., ensure the Japanese label is consistent. For example, if the original indicates country of origin or best-before date, the Japanese label must present correct information as well. If the original display could mislead, consider covering it with the sticker to prevent confusion.
Standardize the format
Use a consolidated labeling panel that groups the items together, placed appropriately and sized so it is readable. Choose a font size and style that prioritizes legibility and does not become too small to read.
Clearly show the importer’s information
For example: “Importer: ○○ Co., Ltd., (address…)” Ensure the responsible Japanese business operator is clearly identified even if the overseas manufacturer is also listed.
Practical points for country-of-origin labeling
As explained, the country of origin is, in principle, the country where the final processing step was completed. However, complex supply chains can make “final processing” unclear. Below are typical cases and how to handle them.
Case 1: Ingredient sourcing country differs from manufacturing country
Example: A dog food made in Japan using chicken sourced from New Zealand.
The final processing/manufacturing is in Japan, so the origin is “Japan” (domestic). If you want to emphasize the ingredient origin, you may add a factual note elsewhere, such as “Made with New Zealand chicken.” However, you must avoid layouts or expressions that could cause consumers to mistake the product as being manufactured in New Zealand (e.g., large flag graphics or oversized “NEW ZEALAND” text).
Case 2: Manufacturing country differs from final packaging country
Example: Product manufactured in Thailand, then finally re-packed into smaller packs in Japan.
Packaging alone generally does not change the essential nature of the product. If the product is already completed in Thailand, the origin remains “Thailand,” even if repacked in Japan. You do not need to indicate the final packaging country. The label should state “Country of origin: Thailand,” and the Japanese business operator information will clarify responsibility in Japan.
Case 3: Multi-stage processing across multiple countries
Example: Ingredients processed in the U.S., final finishing completed in Canada, then imported.
In such cases, the country of origin is the final country where the product is completed—in this example, Canada. Intermediate processing countries are not shown. If absolutely necessary, you may add a small factual note such as “Some ingredients are processed in the U.S.,” but in general, adding multiple country names creates confusion and should be avoided.
If you are unsure, think: “Where did it become the finished product?” Simply filling a bag or can does not necessarily constitute “becoming the product.” The country where the ingredients are mixed, formed, heated/cooked, and turned into the final dog/cat food product is the origin.
Prohibited expressions and key labeling pitfalls
Even if required items are present, inappropriate expressions defeat compliance and trust. Under the Act against Unjustifiable Premiums and Misleading Representations (commonly “the Act” behind misleading advertising rules), and the Fair Competition Code, the following types of unfair or misleading labeling must be avoided. Packaging design and copy should be carefully checked.
Misleading classification or purpose
Do not label a product in a way that could be mistaken for “complete and balanced” or “therapeutic diet” when it does not fit. For example, if a product is not a staple diet but the front panel only says “○○ food (all ages),” consumers may misunderstand it as complete and balanced. Ensure purpose labeling matches the actual product category.
Exaggerated ingredient representation
If a specific ingredient is included only in a small amount, do not emphasize it as if it were a main ingredient. The Fair Competition Code states that terms like “beef,” “chicken,” or “salmon” may not be displayed in the product name or via photos unless that ingredient makes up at least 5% of the contents.
For example, if beef-derived ingredients are only 1%, it is not acceptable to show a large cow photo or “contains beef” prominently. If it is below 5%, use a flavor expression such as “beef flavor” instead.
Unsubstantiated superiority claims
Expressions such as “premium,” “highest quality,” or “No.1” are not allowed unless supported by objective evidence and clear criteria. Ranking claims such as “#1” are only acceptable when based on verified awards or statistical data. Avoid vague copy that merely suggests superiority; use concrete, factual statements where needed.
Defaming competitors or alarming consumers without basis
Do not make claims that unfairly disparage other companies/products or create fear without scientific evidence, such as “○○ company’s food is dangerous” or “grain-containing food is harmful.” Even when comparing products, comparisons must be factual and clear. Internal comparisons such as “X% fewer calories than our previous product” may be acceptable, but claims like “safer than other companies” are not.
False claims about awards or endorsements
Do not display awards, approvals, or recommendations that do not exist. If you use statements like “Winner of ○○ award” or “Recommended by △△ association,” they must be true and verifiable. Fake endorsements are strictly prohibited.
Misleading claims about ingredients or manufacturing methods
Avoid claims that could make consumers believe the product is significantly superior without objective evidence, such as “our proprietary method makes it more nutritious than others.” When promoting manufacturing methods or ingredient features, avoid absolute or comparative claims and keep wording factual and moderate (e.g., “Processed using ○○ method to help maintain freshness”).
Overpackaging (misleading package size)
While not strictly a “label wording” issue, overpackaging can still be misleading. Using packaging far larger than the contents in a way that makes consumers think there is more product may be considered a misleading representation. Use packaging sizes that do not create misunderstanding about the actual net amount.
These NG items are minimum standards under the Fair Competition Code and consumer protection laws. Always conduct multi-person reviews and compliance checks at the design/copy stage. When necessary, consult legal professionals or industry guidelines to confirm the appropriateness of expressions.
Example label format (consolidated labeling panel)
Below is a sample consolidated labeling format for reference:
Name: ○○ pet food (for dogs / adult dogs)
Best before: December 31, 2025 (store at room temperature away from direct sunlight)
Ingredients: Grains (corn, rice), meats (chicken, beef), legumes (soybeans), minerals (calcium, phosphorus), vitamins (A, B2, D), antioxidant (mixed tocopherols)
Country of origin: Kingdom of Thailand
Importer: △△ Trading Co., Ltd., 1-2-3 ○○, Shinjuku-ku, Tokyo
----------------------------------------- -------
Purpose: Complete and balanced dog food (adult dogs)
Net content: 3 kg
Feeding method: Adult dog (5 kg)… approx. 100 g per day (feed in 2 or more meals)
Adult dog (10 kg)… approx. 160 g per day (feed in 2 or more meals)
Guaranteed analysis: Crude protein 24% min, crude fat 12% min, crude fiber 4% max, crude ash 8% max, moisture 10% max
Metabolizable energy (ME): approx. 350 kcal/100 gIn this example, the top section lists the five legally required items (name, best-before date, ingredients, country of origin, and business operator name/address) in Japanese, and the section below the dashed line lists the Code-based items (purpose, net content, feeding method, and guaranteed analysis). ME is shown as a voluntary item.
This is only one example. In practice, the format may vary depending on the product. For example, ingredient listing can be shown by category using parentheses (as in the example) or as a simple descending-order list. Purpose and feeding-method details also differ depending on whether the product is complete and balanced, a snack, etc. Ensure that your product classification and label information match without omissions.
Final checklist for label creation and review
Below is a practical checklist for creating and verifying pet food labels. Checking each point helps reduce the risk of missing items or using inappropriate expressions.
Legal mandatory five items
Fair Competition Code items (industry standard)
NG expressions and risk checks
General practical checks
Incorrect labeling can result not only in violations of consumer protection rules or the Fair Competition Code, but also in penalties under the Pet Food Safety Act (up to one year of imprisonment and/or a fine of up to 1,000,000 yen).
Pet food labeling is not merely a compliance task—it is also an important communication tool that conveys reliability and builds consumer trust. Alongside legal compliance, aim for labels that are clear, consumer-friendly, and honest. Proper labeling supports safer, more trustworthy products that contribute to the well-being of pets and their owners.
Related article: AAFCO guidelines as the basis for complete and balanced foods for dogs and cats.